
Agricultural News
Bob LeValley Offers Beef Quality Assurance Program Injection Site Guidelines
Wed, 25 Aug 2021 09:06:24 CDT
 Weekly, Oklahoma Beef Quality Assurance Coordinator Bob LeValley offers his expertise on animal health. This is a part Oklahoma State University's weekly series known as the "Cow Calf Corner" published electronically by Paul Beck. Today, LeValley talks about injection best practices.
Weekly, Oklahoma Beef Quality Assurance Coordinator Bob LeValley offers his expertise on animal health. This is a part Oklahoma State University's weekly series known as the "Cow Calf Corner" published electronically by Paul Beck. Today, LeValley talks about injection best practices.
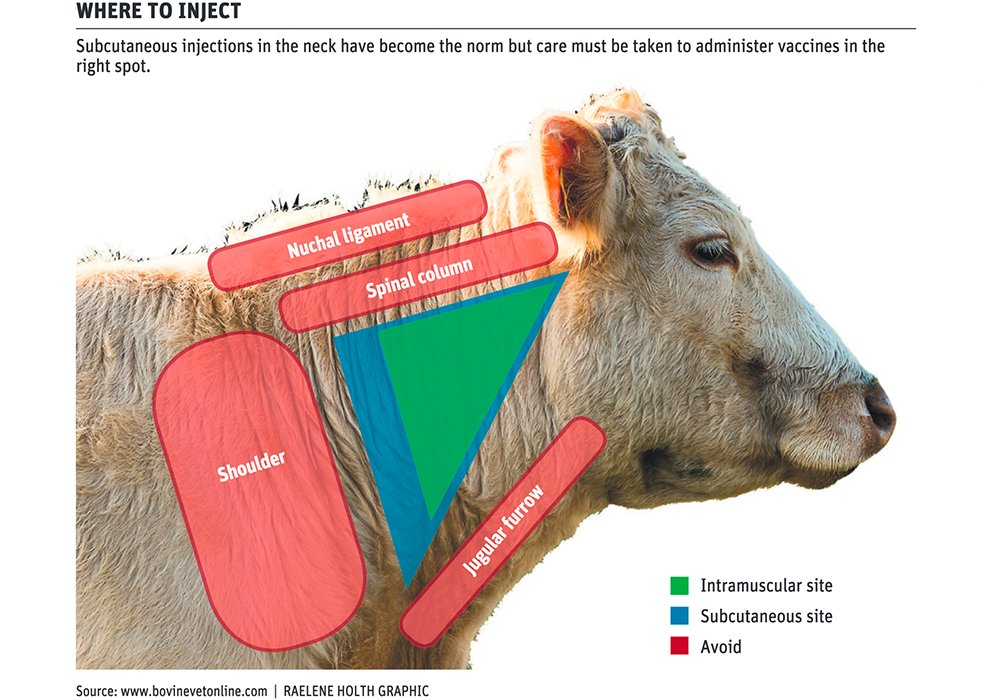
"In order to reduce the incidence of injection site lesions, injectable products should be administered subcutaneous (SQ) if the label allows. Intramuscular (IM) injections not only increase soreness compared to subcutaneous injections, some products given IM cause muscle damage which subsequently causes a significant amount of expensive carcass trim. Knots or blemishes from SQ injections are much easier to find, examine and remove at the packers. Because of these considerations, the national Beef Quality Assurance program adopted a policy that all injections (antibiotics, vaccines, parasiticides, vitamins, prostaglandins, hormones, and all other injectables) be given in front of the slope of the shoulder, that products with SQ labeling be selected in preference to products labeled for IM use only, and that IM injections if required, be limited to not more than 10 cc per injection site."
Producer's Guide for Judicious Use of Antimicrobials in Cattle
Prevent problems: Emphasize appropriate husbandry and hygiene, routine health examinations, and vaccinations.
Select and use antibiotics carefully: Consult with your veterinarian on the selection and use of antibiotics. Have a valid reason to use an antibiotic. Therapeutic alternatives should be considered prior to using antimicrobial therapy.
Avoid using antibiotics important in human medicine as first line therapy: Avoid using as the first antibiotic those medications that are important to treating strategic human or animal infections.
Use the laboratory to help you select antibiotics: Cultures and susceptibility test results should be used to aid in the selection of antimicrobials, whenever possible.
Combination antibiotic therapy is discouraged: Unless there is clear evidence the specific practice is beneficial, select and dose an antibiotic to affect a cure.
Avoid inappropriate antibiotic use: Confine therapeutic antimicrobial use to proven clinical indications, avoiding inappropriate uses such as for viral infections without bacterial complication.
Treatment programs should reflect best use principles: Regimens for therapeutic antimicrobial use should be optimized using current pharmacological information and principles.
Treat the fewest number of animals possible: Limit antibiotic use to sick or at-risk animals.
Treat for the recommended time period: To minimize the potential for bacteria to become resistant to antimicrobials.
Avoid environmental contamination with antibiotics: Steps should be taken to minimize antimicrobials reaching the environment through spillage, contaminated ground run off or aerosolization.
Keep records of antibiotic use: Accurate records of treatment and outcome should be used to evaluate therapeutic regimens and always follow proper withdrawal times.
Follow label directions: Follow label instructions and never use antibiotics other than as labeled without a valid veterinary prescription.
Extra-label antibiotic use must follow FDA regulations: Prescriptions, including extra label use of medications must meet the Animal Medicinal Drug Use Clarification Act (AMDUCA) amendments to the Food, Drug, and Cosmetic Act and its regulations. This includes having a valid Veterinary/Client/ Patient Relationship (VCPR).
WebReadyTM Powered by WireReady® NSI
Top Agricultural News
More Headlines...





